JAMB Chemistry Questions and Answers Expo Runz 2024/2025
This platform Expocoded.com has been created to help all JAMB candidates with Chemistry Expo, questions and answers. It is no longer news that candidates make use of Expo in the JAMB utme examination.
Unless you want to fail or personally don’t support Expo, this opportunity is open to everyone. Our Expo is for all candidates who want to liberate themselves from JAMB bondage.

Are you wondering if the JAMB Chemistry syllabus for the 2024 session has been released? Stay informed about this year’s JAMB Chemistry syllabus and its release date. Gain insights into the topics that JAMB frequently includes in Chemistry questions and discover the most repeated topic in the JAMB Chemistry exam. For accurate and up-to-date information, visit the official JAMB website or contact JAMB directly.
Hello, you are highly welcome to Jambexpo.com Legit JAMB Examination help centre. You are welcome once again to the 2024 jamb runz portal. We offer you the best jamb runz service. With our direct to system package, you can score above 300 and with our normal system package, you can score above 280+
We assure you the best. We are trusted and tested 💯
THOSE WHO REGISTERED IN OUR DATABASE will be granted access to all their personal questions and answers
- Now, let me introduce you fully to the program. JAMB Chemistry Questions and Answers 2024 Expo Runz is now available at EXPOCODED.COM is a known Expo and News forum where you can get sure 2024 JAMB Expo, NECO 2024 expo, 2024 WAEC QUESTIONS AND ANSWERS NABTEB expo 2024, IJMB, JUPEB assistance. For the 2024 jamb EXPO, you will need to subscribe if you want to get the questions and answers before the exam.
Expocoded.com greatest joy is for you to pass your 2024 JAMB Chemistry examination with ease because reading is the key to success, try your best, so you can be able to defend yourself and your certificate anywhere, please note we only help to back you up and our backup is certain and solidify with 100% assurance as you have to subscribe for it by following the Procedure below.
RECOMMENDED LINKS
- 2024 JAMB CBT EXPO
- JAMB CBT AREAS OF CONCENTRATION
- JAMB CBT SUBJECT COMBINATION
- JAMB CBT SUBJECT SYLLABUS FOR ALL SUBJECT
- JAMB CBT LIKELY QUESTIONS AND ANSWERS FOR ALL SUBJECT
- JAMB CBT REGISTRATION CENTRE
- JAMB Brochure
- JAMB English Language Questions and Answers
- JAMB Mathematics Questions and Answers
- JAMB Chemistry Questions and Answers
- JAMB Chemistry Questions and Answers
- JAMB Biology Questions and Answers
- JAMB Geography Questions and Answers
- JAMB Economics Questions and Answers
- JAMB Literature in English Questions and Answers
- JAMB Government Questions and Answers
- JAMB Commerce Questions and Answers
- JAMB Finacial Accounting Questions and Answers
- JAMB Agric Science Questions and Answers
- JAMB CRS Questions and Answers
- JAMB IRS Questions and Answers
- JAMB Yoruba Questions and Answers
- JAMB Igbo Questions and Answers
- JAMB Hausa Questions and Answers
- WAEC GCE Examination Expo
- WAEC GCE Registration Guidelines
- WAEC GCE Timetable Second Series
- NECO GCE Registration Guidelines
- NECO GCE External Timetable
- NABTEB GCE Examination Expo
- NABTEB GCE Registration Guidelines
- NABTEB GCE Nov/Dec Exam Timetable
- Login To Jamb Portal Using Registration Number
We don’t just offer Jamb runs or a good Jamb expo site for 2024, we offer 280-320 JAMB SCORE
if you are here directly or indirectly for the 2024 JAMB cbt Chemistry Expo Assistance Program? ARE YOU LOOKING FOR A LEGITIMATE SOURCE FOR YOUR EXAM SUCCESS IN THE 2024 JAMB CBT EXPO? Jambexpo.COM IS THE MOST RELIABLE 2024 JAMB EXPO (RUNZ/EXPO) WEBSITE FOR YOU How to get JAMB Latest Exam 2024 – Correct Legit 2024 JAMB Expo Exam | Receive Answers 6 hours or direct upload to your exam system, no delay… And also remember here Is the Only Place to get Legit, Correct and Verified sources for the upcoming All Subjects – 2024 JAMB cbt Expo Questions And Answers Exam Expo Runz On.
JAMB examination is coming up soon now, and our main duty is to help all Great Nigeria students. We remain your powerful legit source for you to excel and make 280+ exams with flying colours with just a sitting and say Bye Bye to Ye Ye JAMB Exam forever.
Expocoded.com JAMB Chemistry Questions and Answers 2024/2025 Expo Runz are from certified legit sources, and with our special VIP treatment for early subscribers, you know that you have the best assistance in your JAMB CBT EXAM. We don’t just offer Jamb exam runs or a good Jamb expo site for 2024, we offer 280-320 JAMB SCORE. PLEASE DON’T FALL INTO ALL THESE NEW WEBSITES THAT DO NOT HAVE SOURCES, WE ARE OLD IN THIS BUSINESS, SUBSCRIBE WITH US FOR A BETTER RESULT IN YOUR JAMB 2024 (EXPO/RUNS)
We are the best 2024 JAMB cbt Expo website and legit jamb expo runz website in Nigeria. in other to know we offer the best jamb runz and jamb 2024 expo, jamb cbt runs, jamb examination questions, and answers. Those who subscribed with us last year and other years’ exams can testify how we upload their jamb questions and answers directly to their computer because they registered in our database using our jamb 2024 access code
For your success to count through, You need to sacrifice whatever it is to succeed. Those charging you 4,000 for JAMB Runs are nothing but Sourceless copycats that will still come out boasting their own opinion.
I speak less and do more and drop evidence of testimony in bags.
I repeat Jamb Runs isn’t run by anyone randomly, So be careful in picking your choice. I’m Not forcing you to subscribe with me But be careful while choosing.
Remember Cheaper product sees many buyers while the original Product sees fewer buyers. I assure you nothing less than 280+ for direct mobile while 300 for direct to the system package
Guaranteed. Many Out there will deceive you into scoring 300. So Those Who know what it is to write a jamb twice will Rush Towards a Subscription to avoid stories that touch the Heart.
Here at 2O23 JAMB EXPO CBT, we make the thing happen
Are you wondering if the JAMB Chemistry syllabus for the 2024 session has been released? Stay informed about this year’s JAMB Chemistry syllabus and its release date. Gain insights into the topics that JAMB frequently includes in Chemistry questions and discover the most repeated topic in the JAMB Chemistry exam. For accurate and up-to-date information, visit the official JAMB website or contact JAMB directly.
As the 2024 jamb ended we started looking for a new alternative way to help our candidates who will be taking the much anticipated 2024 jamb Chemistry Questions and answers.
We understand knowing where jamb will set jamb Chemistry Questions will go a long way in helping thousands of candidates perform better.
In last year’s jamb Chemistry we were able to predict where jamb is going to set their questions from, honestly the majority of our candidates who took us serious smile out from the exam hall.
Jamb Chemistry questions and answers
on a yearly basis, Expocoded.com tried as much as it can by giving out key points from which jamb are expected to set their physics questions from.
In this year jamb, we will also show you where the jamb will set their questions from and also provide answers, Expo for them.
how jamb Chemistry Questions Will appear on the computer
for those who have not written a computer base exam, We have provided a past question from 2016 JAMB below.
What’s our aim for you on Jamb Chemistry
We have just one objective here as far as jamb Chemistry is concerned and that objective is to try our possible best to guide you on how to score nothing less than 250+ in this year’s jamb only if you work with us We have the direct system and the SMS package
The direct system, is the package where you provide us with your jamb reg number, and we will log in and answer all your questions for you, so when you get to your center you will see that all your questions have been answered by our team, and the price for it is #15000, with a grantee score of 310 and above,
And the SMS package is when we send the answers to your mobile phone 3hours before your exam, with the same jamb reg number you will provide for us, so you can memorize it before your exam.
The price for this SMS package is #10000, with a grantee score of 280 above.
What will jamb set in Chemistry?
honestly, there will be nothing new in jamb Chemistry Questions, in fact, jamb We see jamb bringing up old questions from previous jamb questions.
those who will be able to read and understand jamb past questions and answers accurately will smile out of the exam hall.
see: how Jamb 2024 will look like
What’s the way forward in jamb Chemistry questions and answers
since we have exposed jamb’s weakest point what is left for you is to go through jamb past questions and answers.
you might ask where can I get the updated version of jamb past questions and answers from? we are going to provide it for you here free of charge all you need is just to have your mobile data on. click here to read on how to read and understand jamb past questions
see: how Jamb’s use of Chemistry will look like
Where can i get jamb past questions and answers on Chemistry from
It’s sold in the market for just 600 to 1000 naira in major bookstores around.
make sure you get the updated version at least from 1985 to 2014. you will hardly see that of 2015 in the market
IMPORTANT INFO ON 2024 MIDNIGHT JAMB EXP0
100% Refund If 2024 Midnight JAMB Expo Is Not Delivered To Your TasTe And Satisfaction. 100% Assurance And Authentication Of You Getting Your Question And Answers In The Night. Don’t Panic We Never Fail To Our Promise. Give It A Try And Enjoy Our Service.
Once Again Dont Be Deceived By Other Sites Or WhatsApp People Or Groups Promising And Telling Stories They will give you Fake Or Late Answers. For Your Good Kindly Subscribe To Us For 100% Assurance.
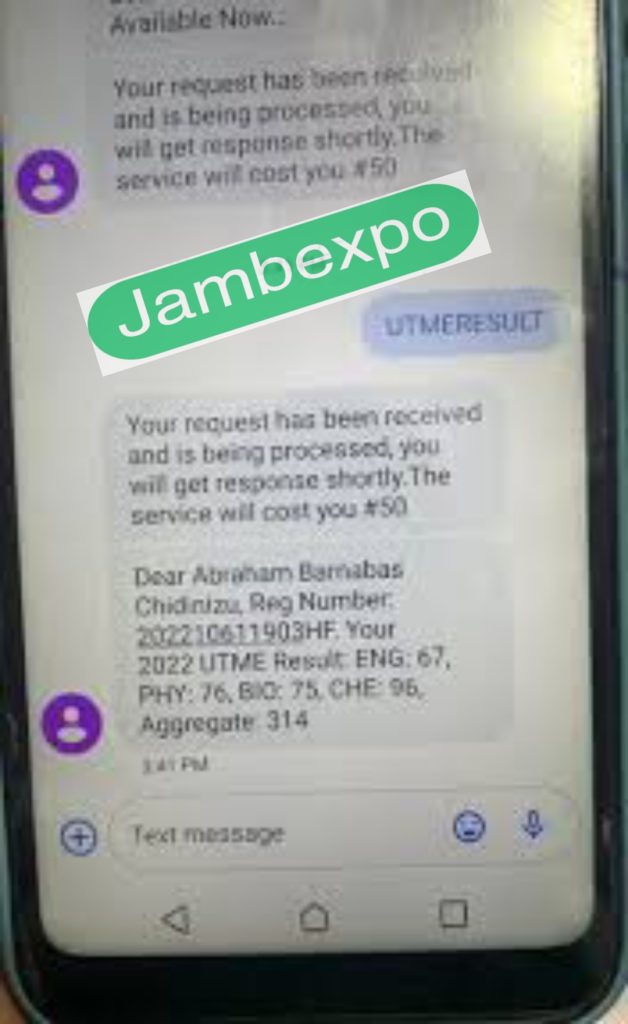
After the Successful use of our device and software called the internal micro uploader (I.M.U) AND (N.L.P) Natural Language Process To upload your jamb questions and answers directly to your computer, see how ur jamb questions will appear the moment u put your jamb registration number on your exam day 👉
Please Watch and see how we upload your answers using our I.M.U AND N.L.P Software
- On the day of your exam, log in to your system with your Jamb Registration Number, at the top right corner of the system you will see a login access code click on it and log in.
This is exactly what you will be like after we are done working on your Registration number, We are trusted and tested by thousands of students who made their results through us
How to Register to Our Online 2024 JAMB Expo Database
- Expocoded.com, Subscribe to your preferred plan to receive an ACCESS CODE. Each ACCESS CODE can ONLY be used by ONE candidate to register to our Database. This means that you cannot share your Access Code with your friends.
- Go to expocoded.com
- Find the candidate registration and click on “Register Here” then follow the prompt.
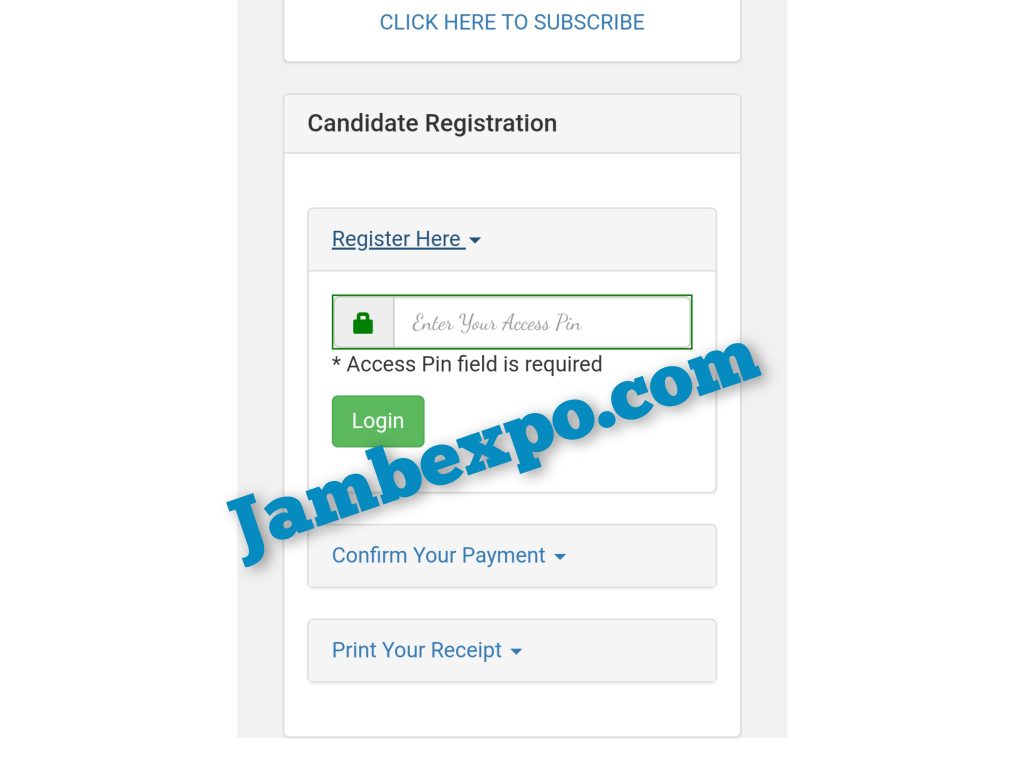
- Input the ACCESS CODE Received to start your Registration.

- Register your details correctly and proceed to the next step.
- Input the JAMB Registration Number and click on “Confirm Payment” to confirm your details.
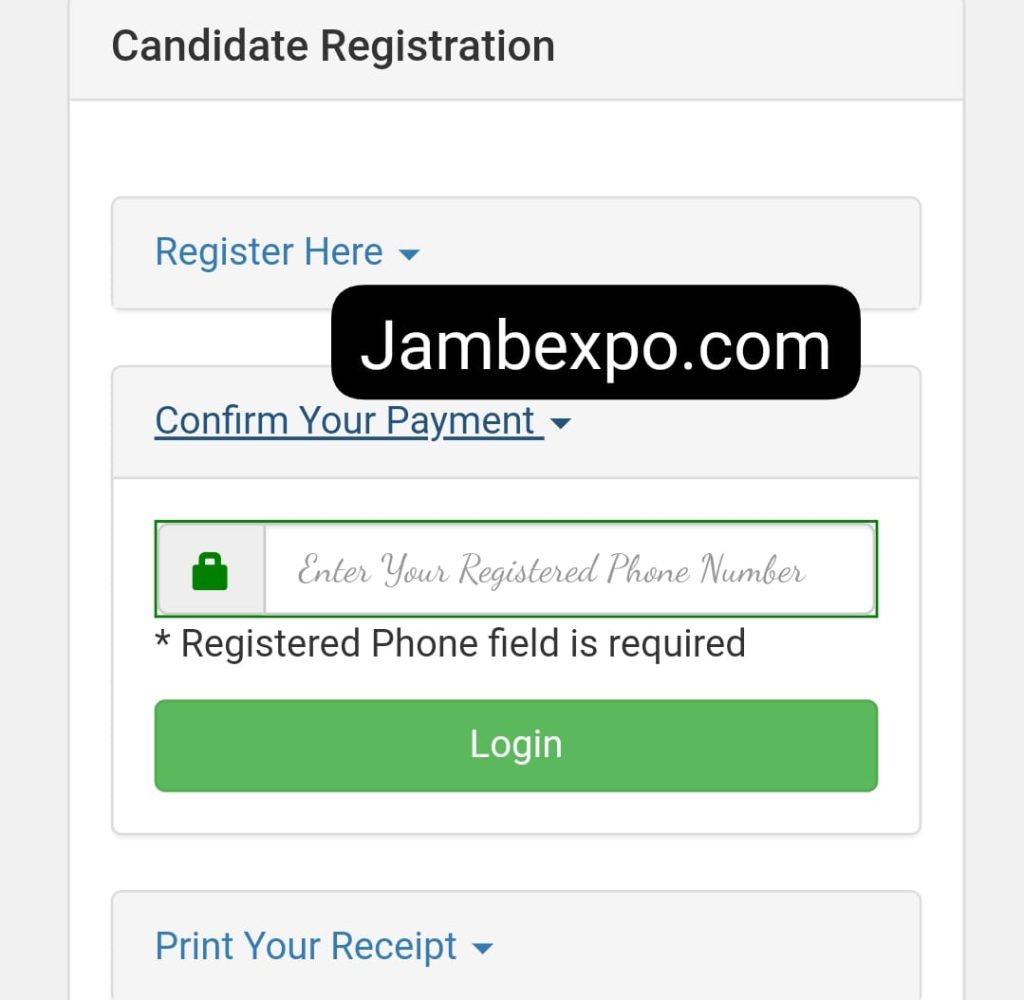
7. Then, after successful registration, you can proceed to “Print Your Receipt” for reference purposes.

NOTE: Only candidates registered to our database will be able to receive their 2024 JAMB UTME Expo Answers before the scheduled examination time. ONCE YOUR PAYMENT IS RECEIVED, A Confirmation Text Message with Your ACCESS CODE to start your Registration Here Will be Sent to Your Phone Number to enable you to Exam code register at portal.jambexpo.com.
How to View Your 2024 JAMB UTME Expo Answers with Your JAMB Registration Number
- Before proceeding, ensure to have Registered at portal.jambexpo.com.
- Visit portal.jambexpo.com.

- Enter your JAMB REGISTRATION NUMBER only.

- Proceed and click on “CHECK” to get your Answers.
IMPORTANT INFO ON 2024 MIDNIGHT JAMB Expo
100% Refund If 2024 Midnight JAMB Expo Is Not Delivered to Your Taste and Satisfaction. 100% Assurance and Authentication of You Getting Your Question and Answers in the Night. Don’t Panic We Never Fail to Our Promise. Give It a Try and Enjoy Our Service.
Once Again Don’t Be Deceived by Other Site or WhatsApp People or Groups Promising and Telling Stories They will give you Fake or Late Answers. For Your Own Good Kindly Subscribe to Us For 100% Assurance.
SEE HOW THE ANSWERS WILL BE ON YOUR SYSTEM
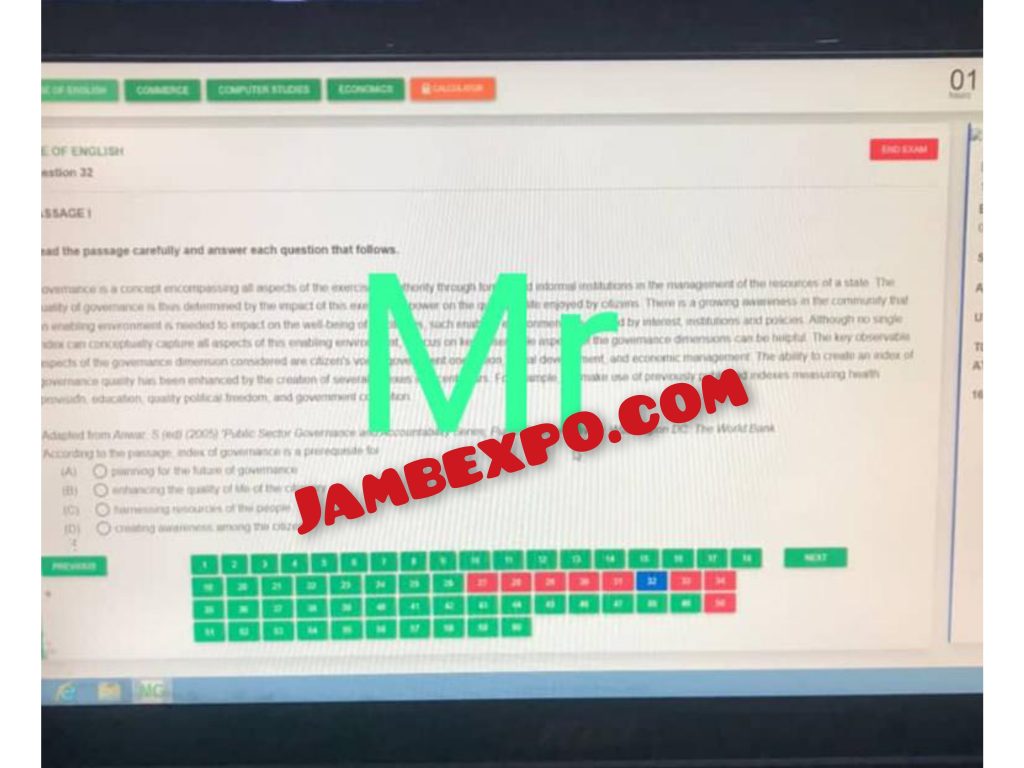
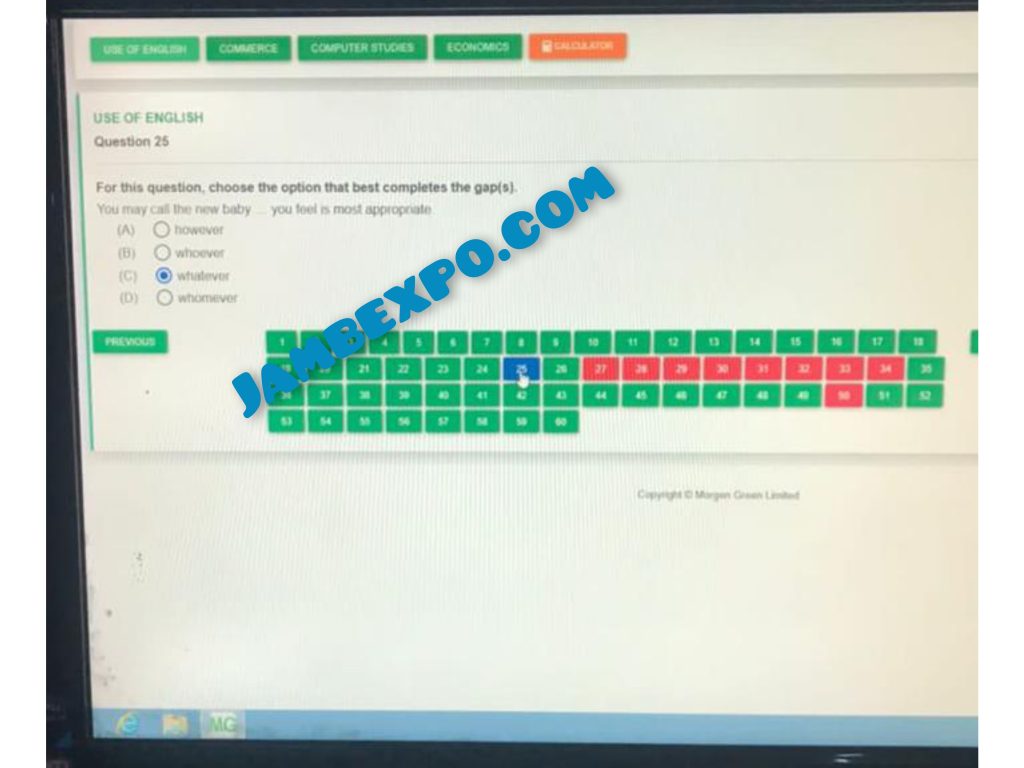
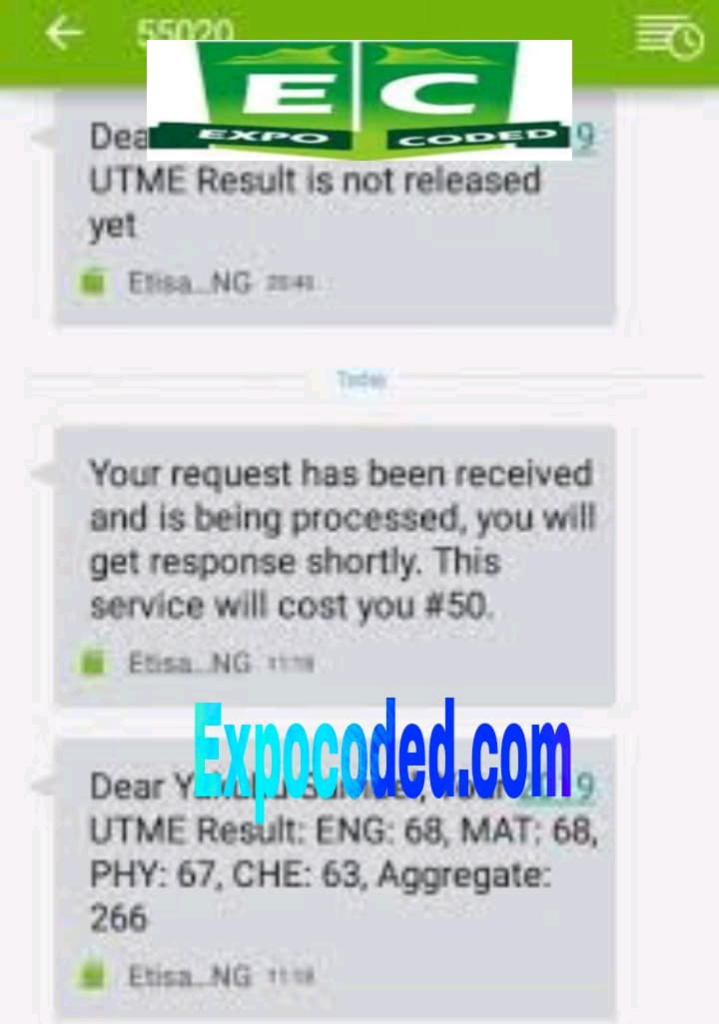
See JAMB Prove of Our Previous Year’s Subscribers




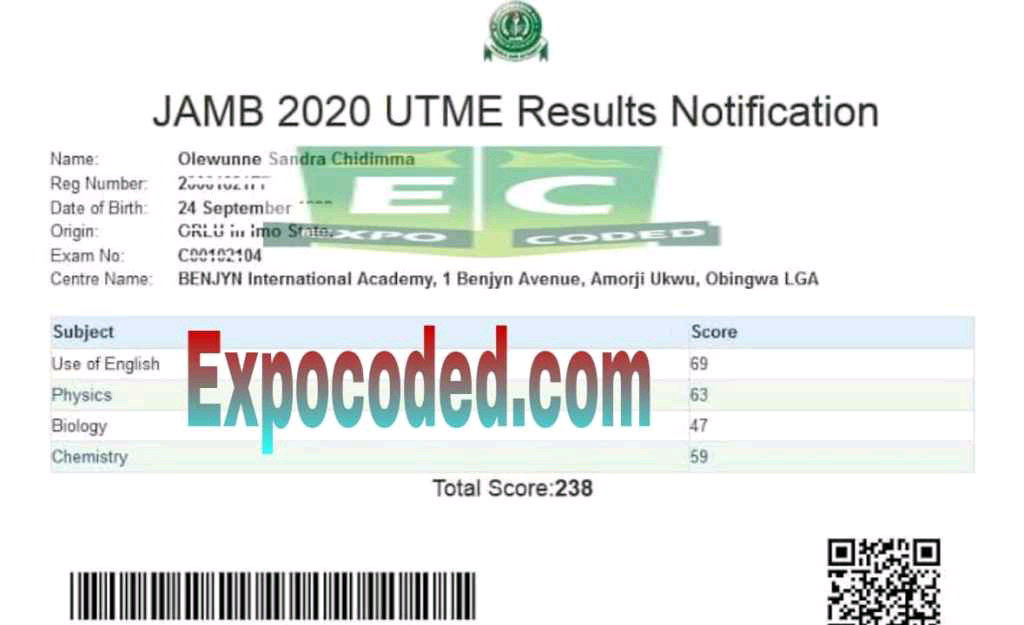
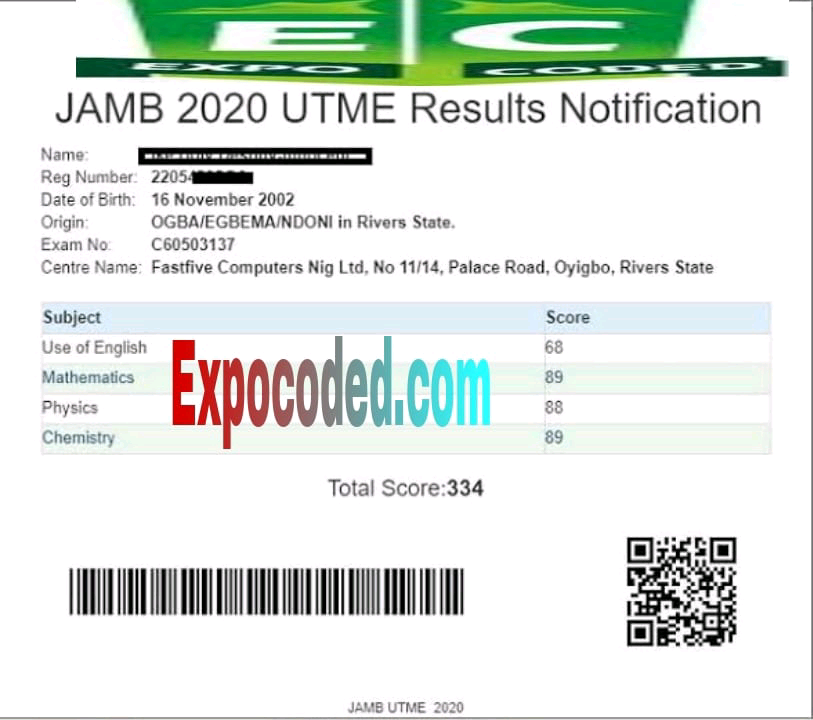



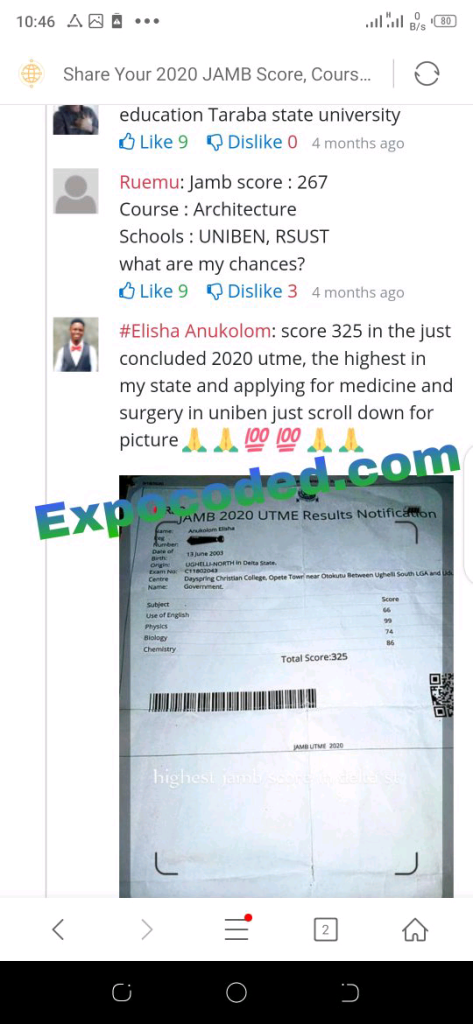
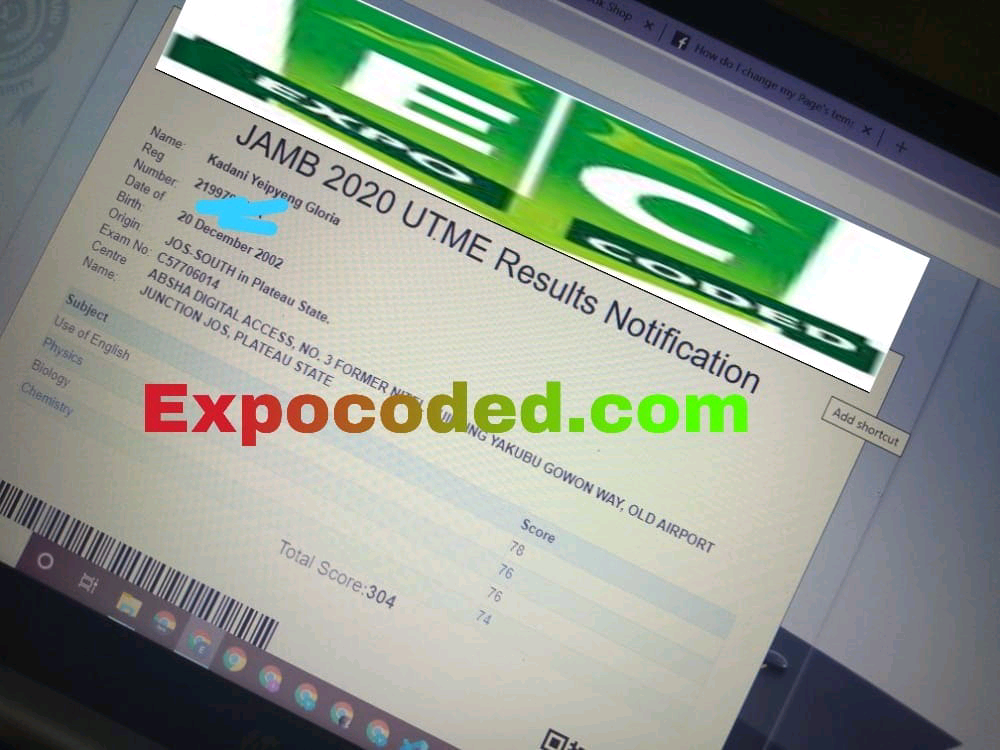

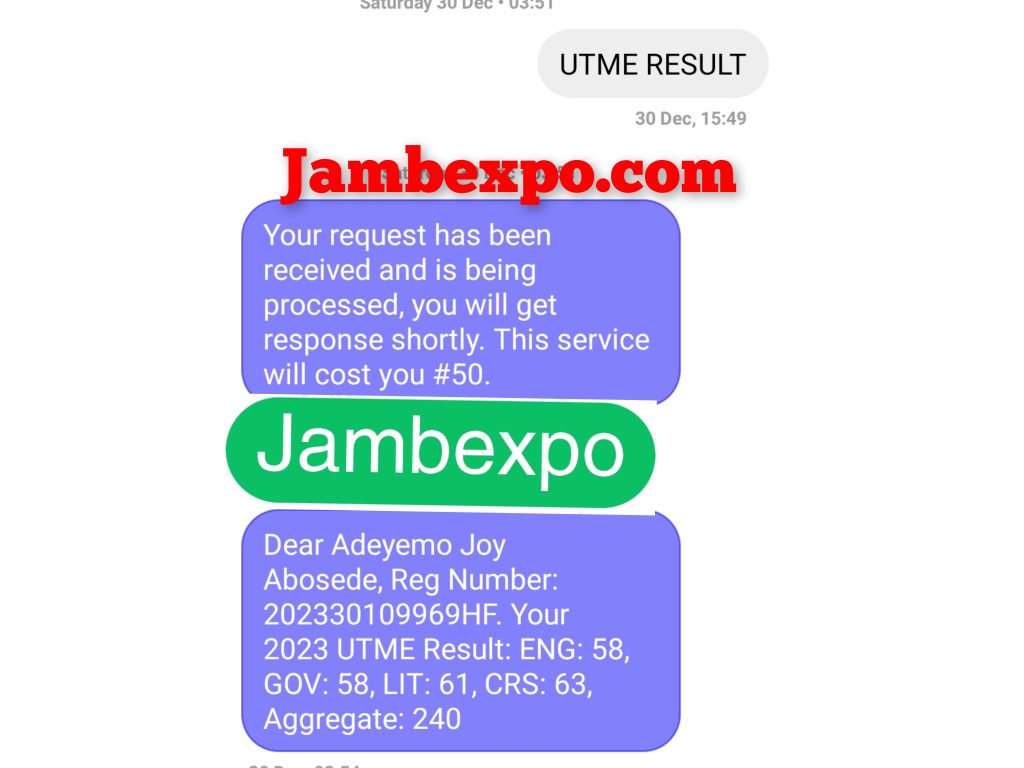
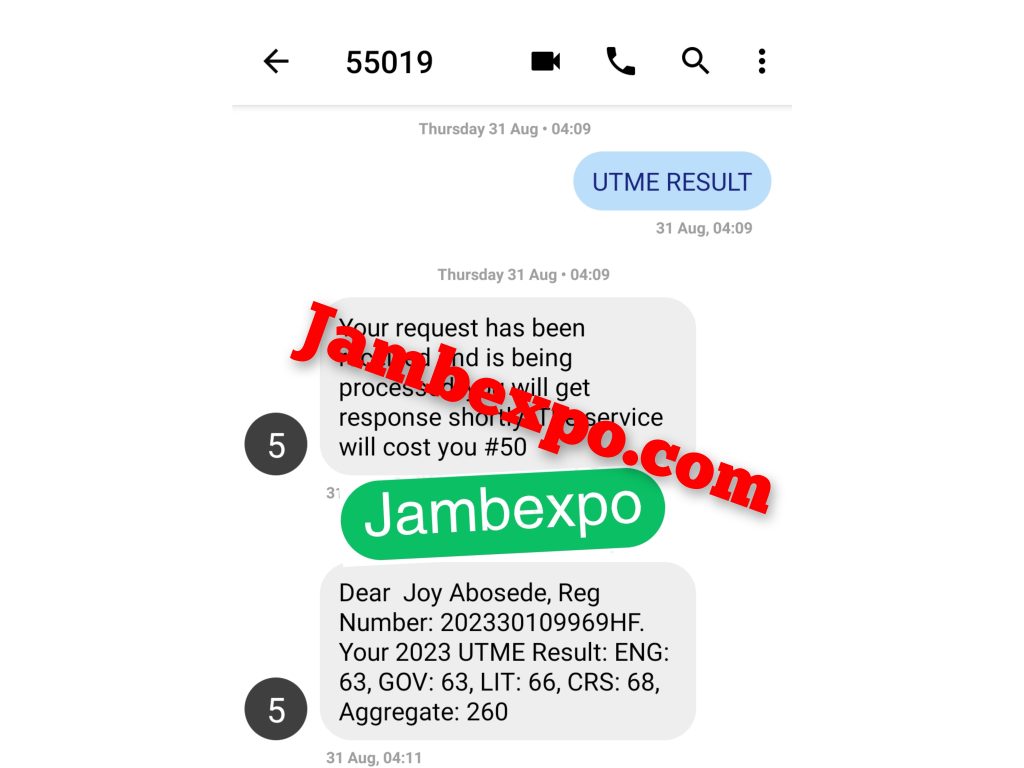
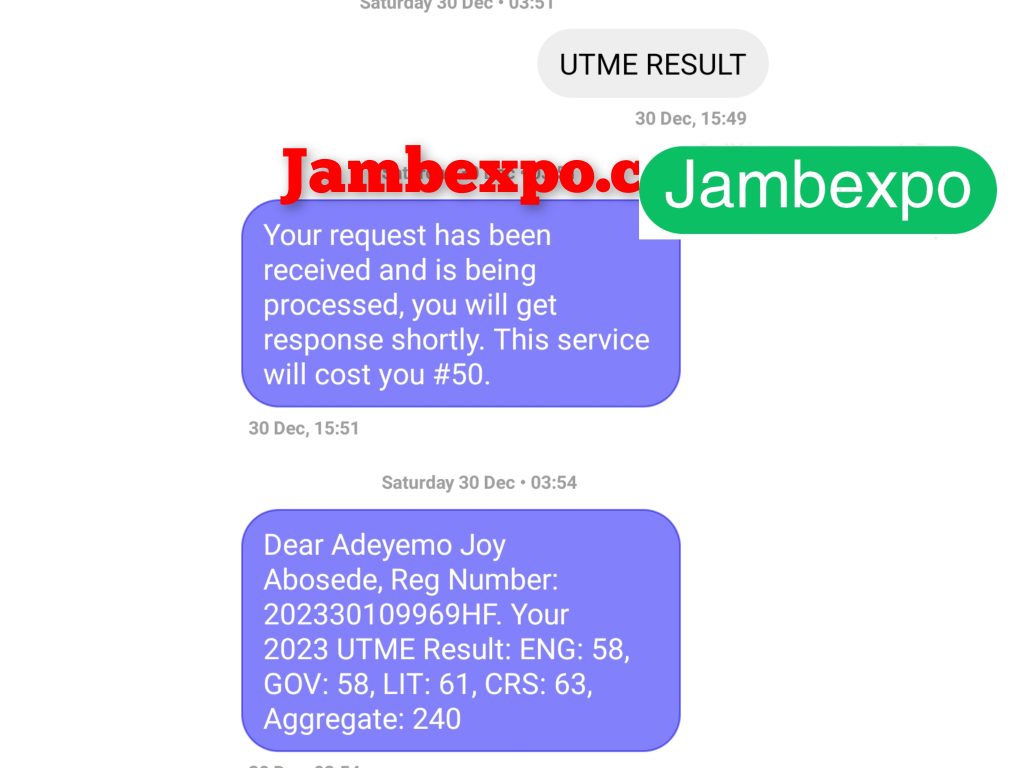
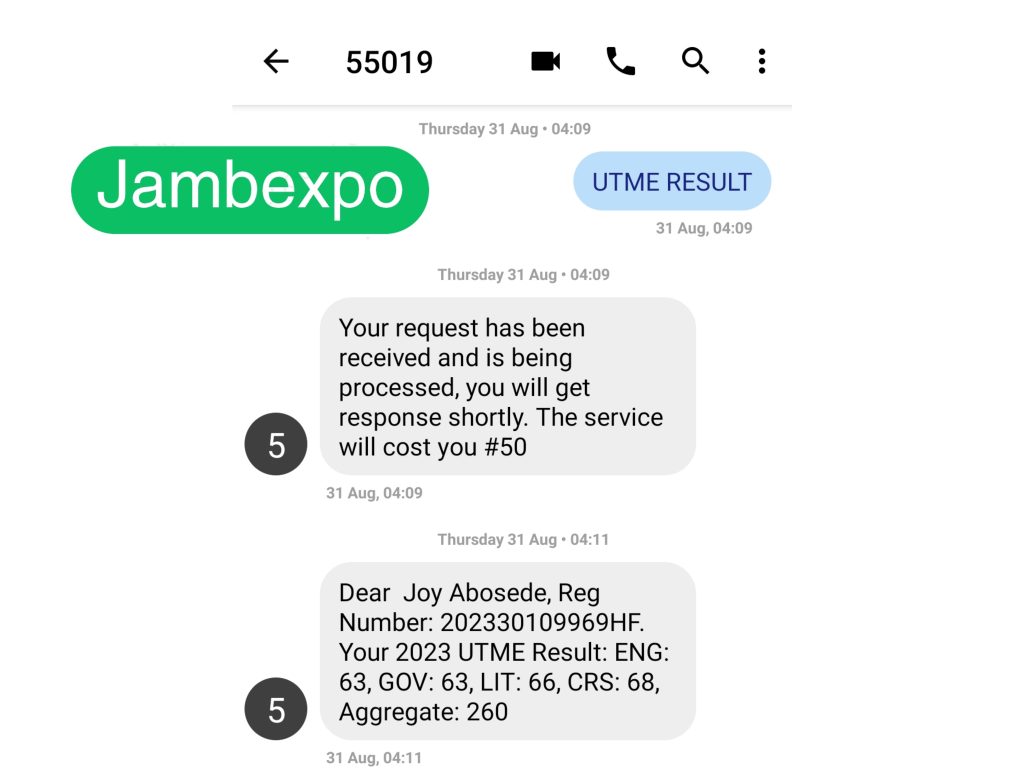

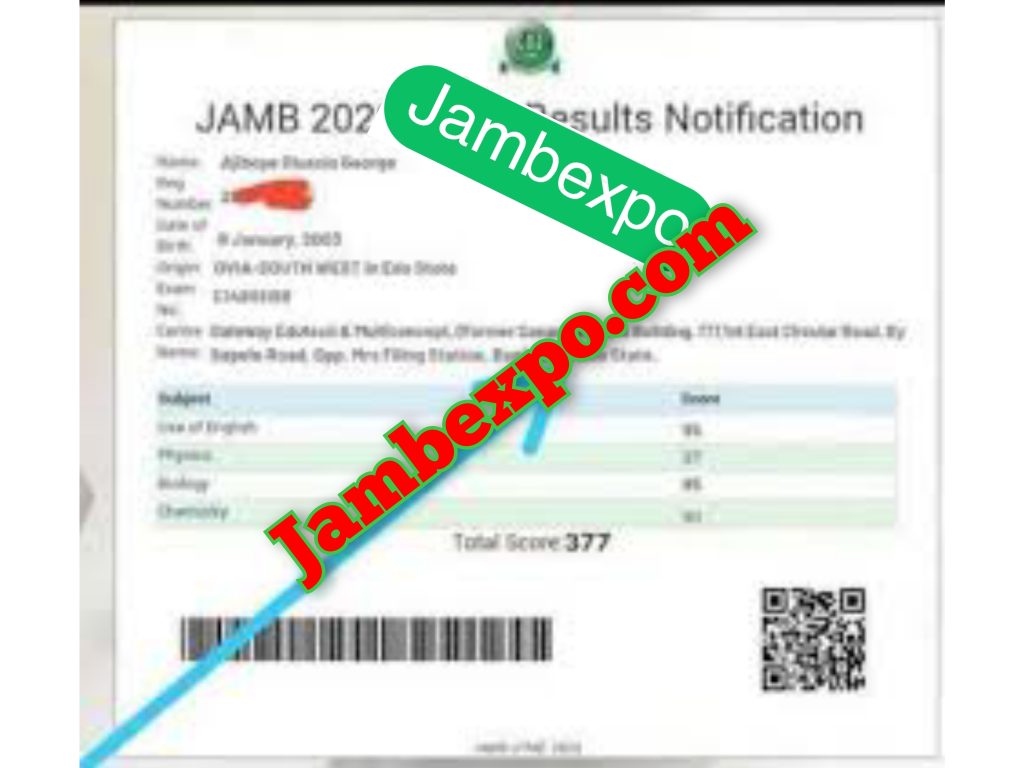
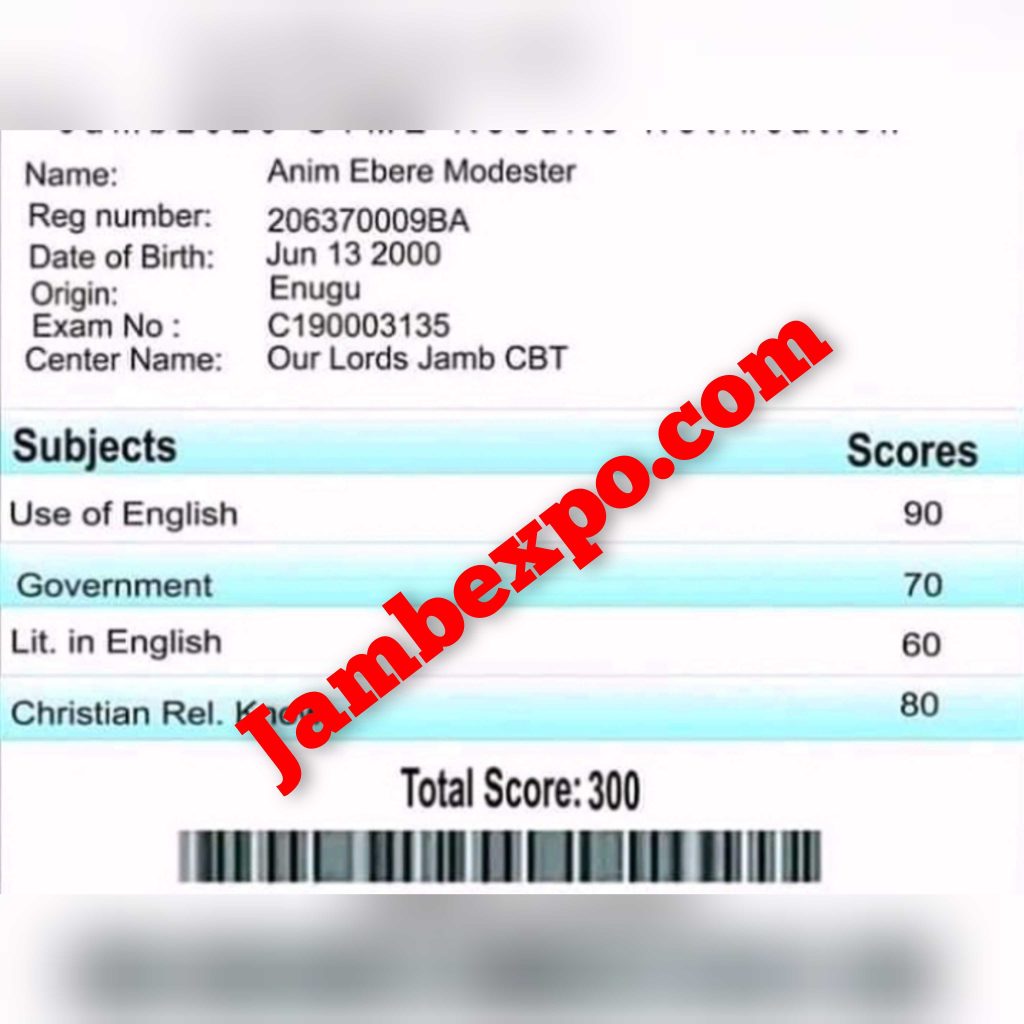
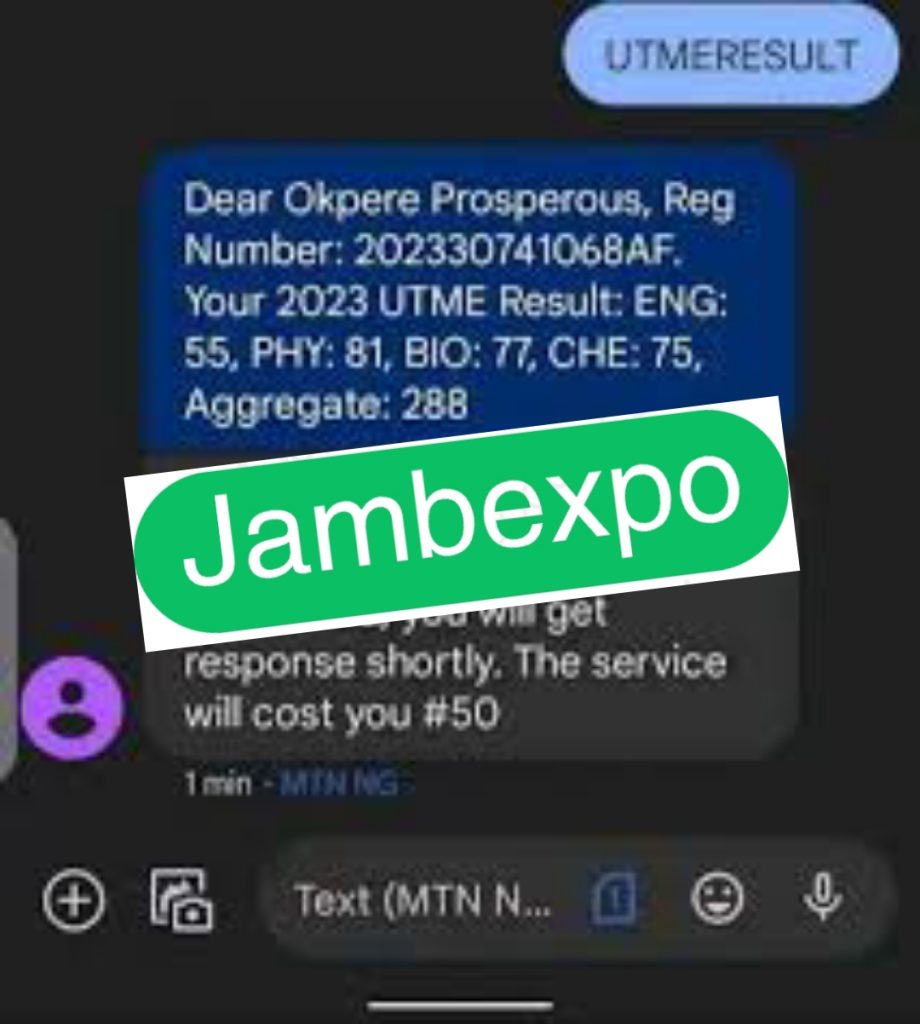
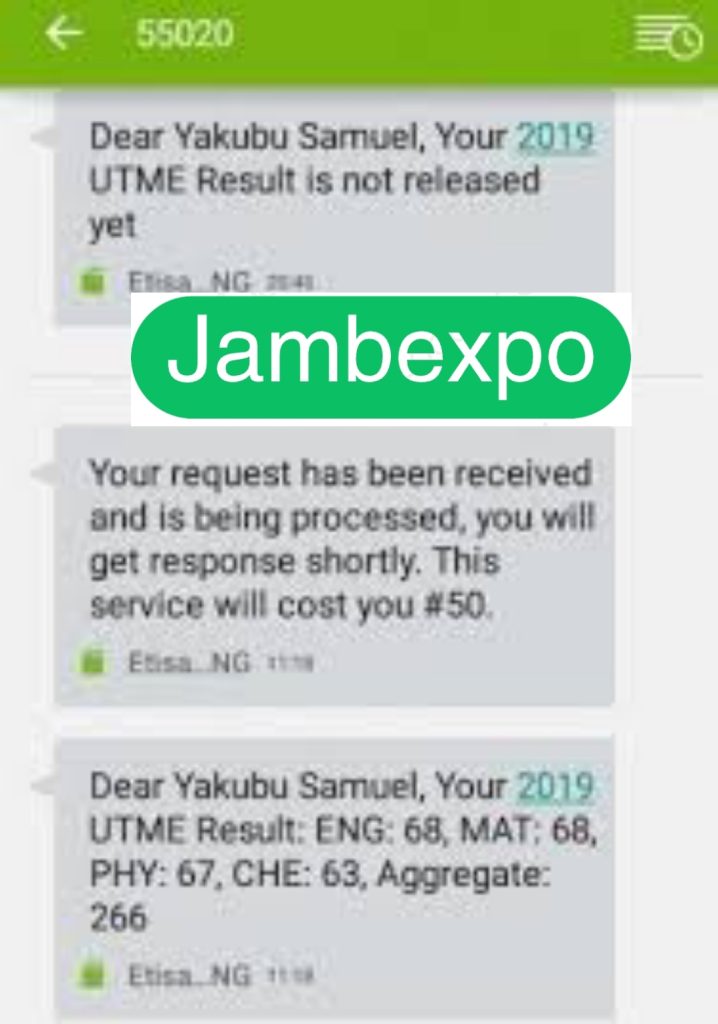
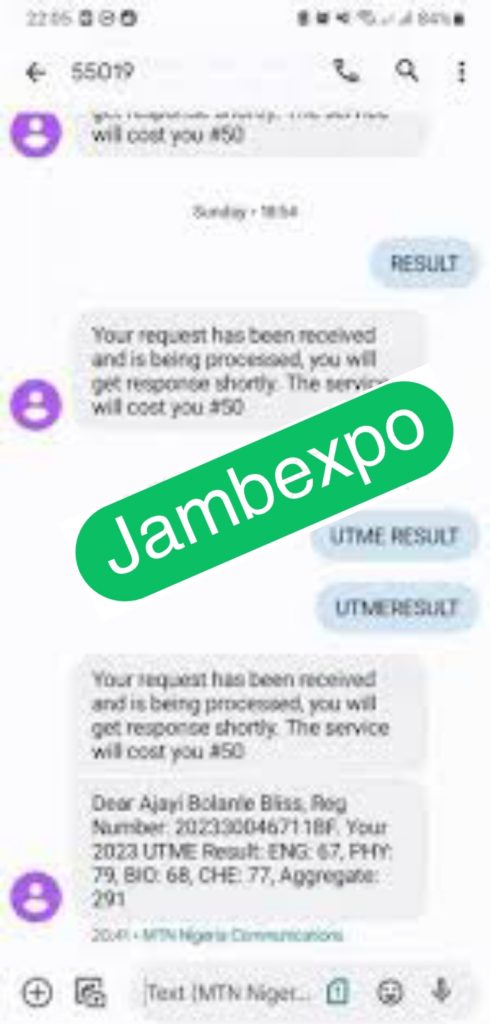

Expocoded.com subscribers’ results
As an Expocoded.com subscriber, you will have access to our CBT Questions and Answers a 6hours before your examination. This will give you ample time to study and understand the topics covered. Our CBT Questions and Answers are compiled by JAMB subject experts. Hence, you can be sure of their authenticity and effectiveness.
We also offer counselling services to our subscribers. Our counsellors are experienced professionals who will guide you on how to approach the examination, what to expect and how to deal with exam anxiety. With our counselling services, you will be more confident and better prepared for the examination.
JAMB Chemistry Questions and Answers Expo Runz, jamb Chemistry expo 2024 , jamb Chemistry runz 2O24, jamb Chemistry runs 2024, jamb Chemistry Answers 2024, jamb Chemistry Questions and answers 2024, jamb 2024 Chemistry Questions and answers, jamb 2024 Chemistry Questions and answers, jamb 2024 Chemistry Questions and answers, 2024 jamb Chemistry expo, 2024 jamb Chemistry expo, 2024 jamb Chemistry expo, 2024 jamb Chemistry expo
jamb 2024 Chemistry Questions and answers, jamb 2024 Chemistry Questions and answers, jamb 2024 Chemistry Questions and Answers, jamb Chemistry Questions and Answers 2024, free jamb expo site,